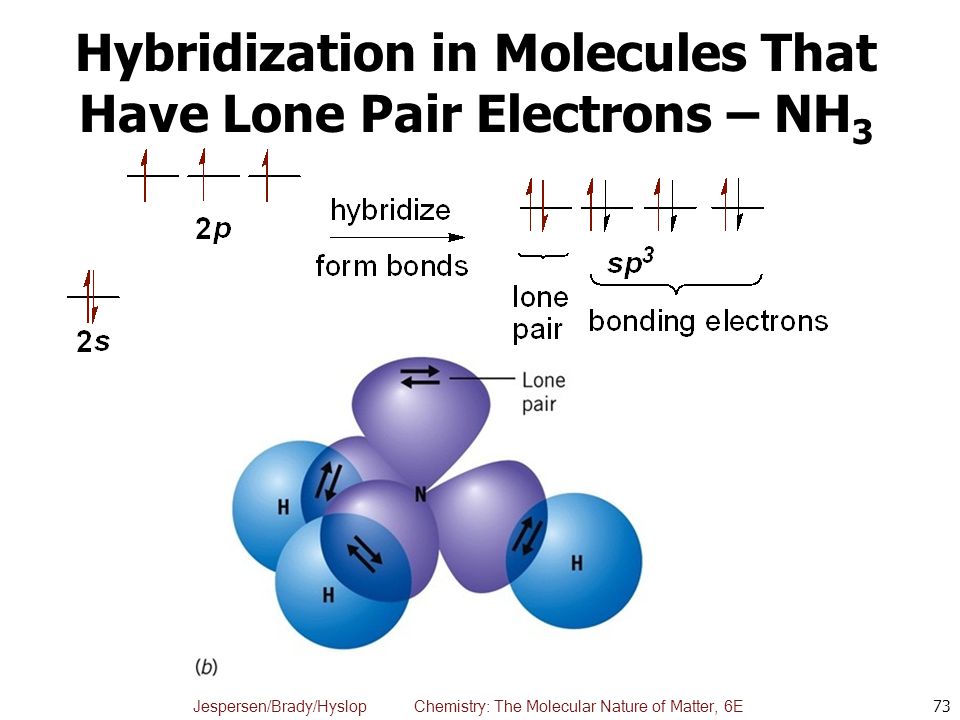
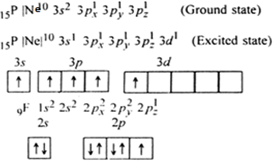
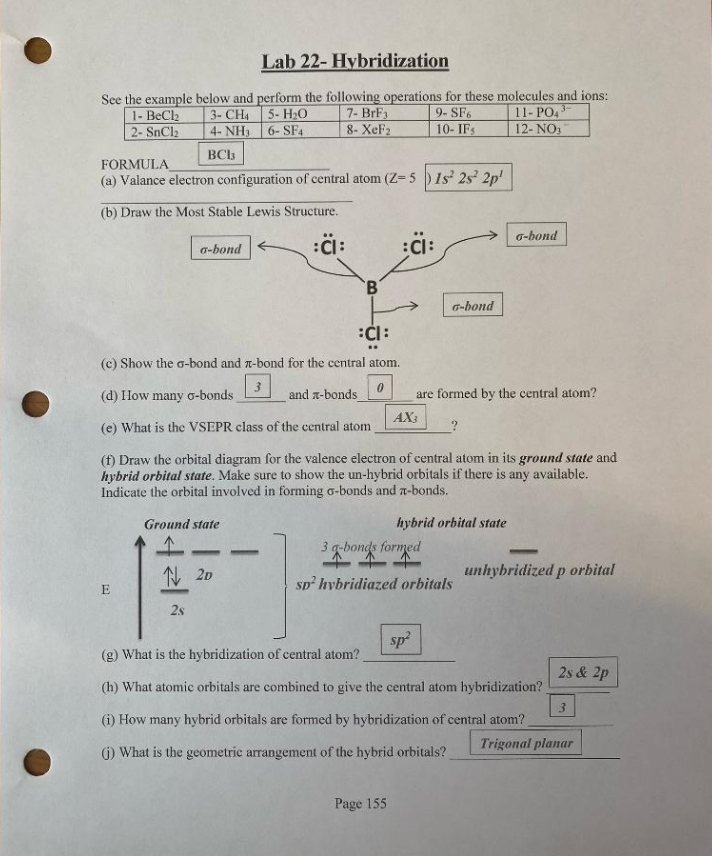
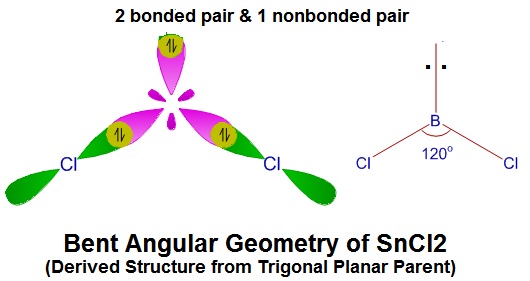
Find hybridisation , shape of the following molecule- SnCl2 SF4 ClF3 ClF5 - Chemistry - Chemical Bonding and Molecular Structure - 16845639 | Meritnation.com
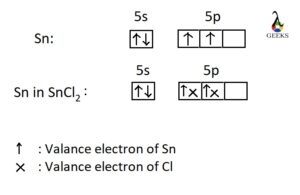
SnCl2 Lewis Structure: Drawings, Hybridization, Shape, Charges, Pair And Detailed Facts - LAMBDAGEEKS

VSEPR theory geometry of sncl2 molecule nh3 mol h2o clf3 xef2molecule xef4 molecules fy bscsemester - YouTube

SOLVED: Under some conditions SnCl2 can simultaneously behave as both a Lewis acid and a Lewis base. Suggest a molecule that might be formed under these conditions. What is the hybridization on