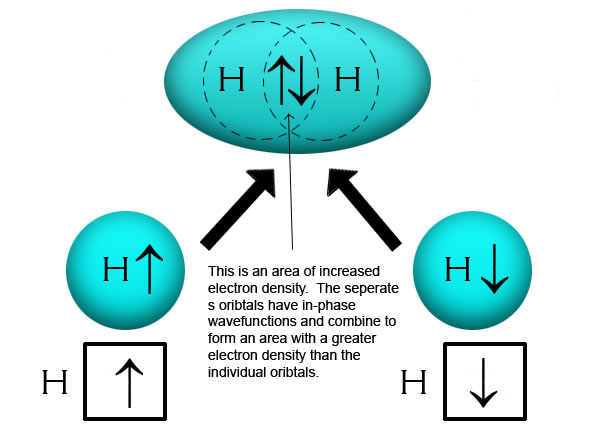
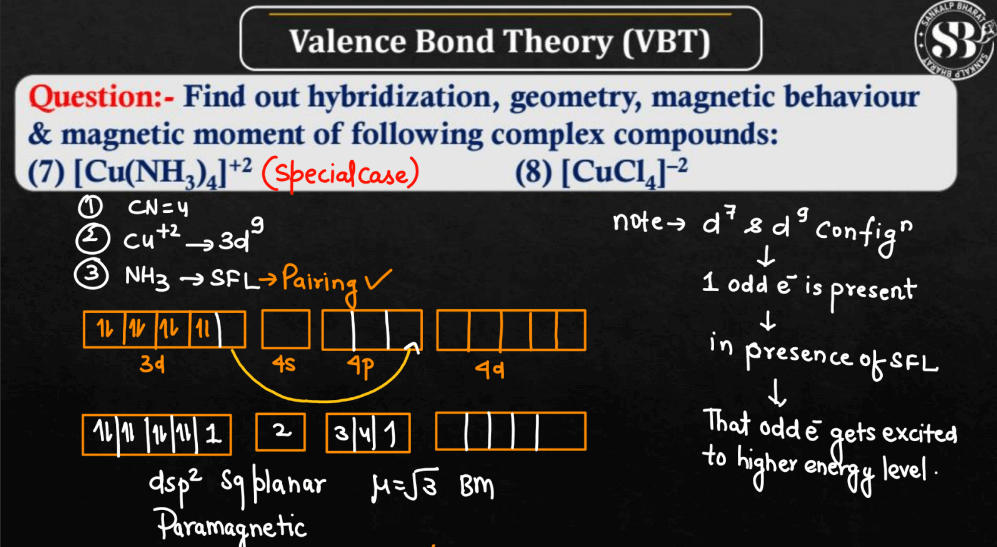
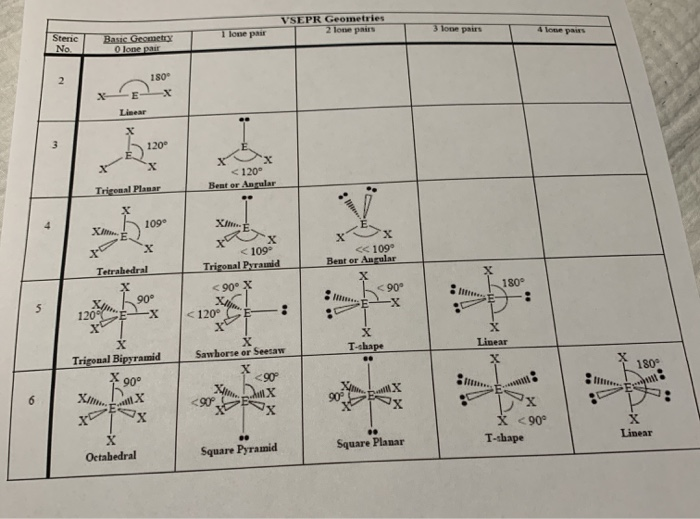
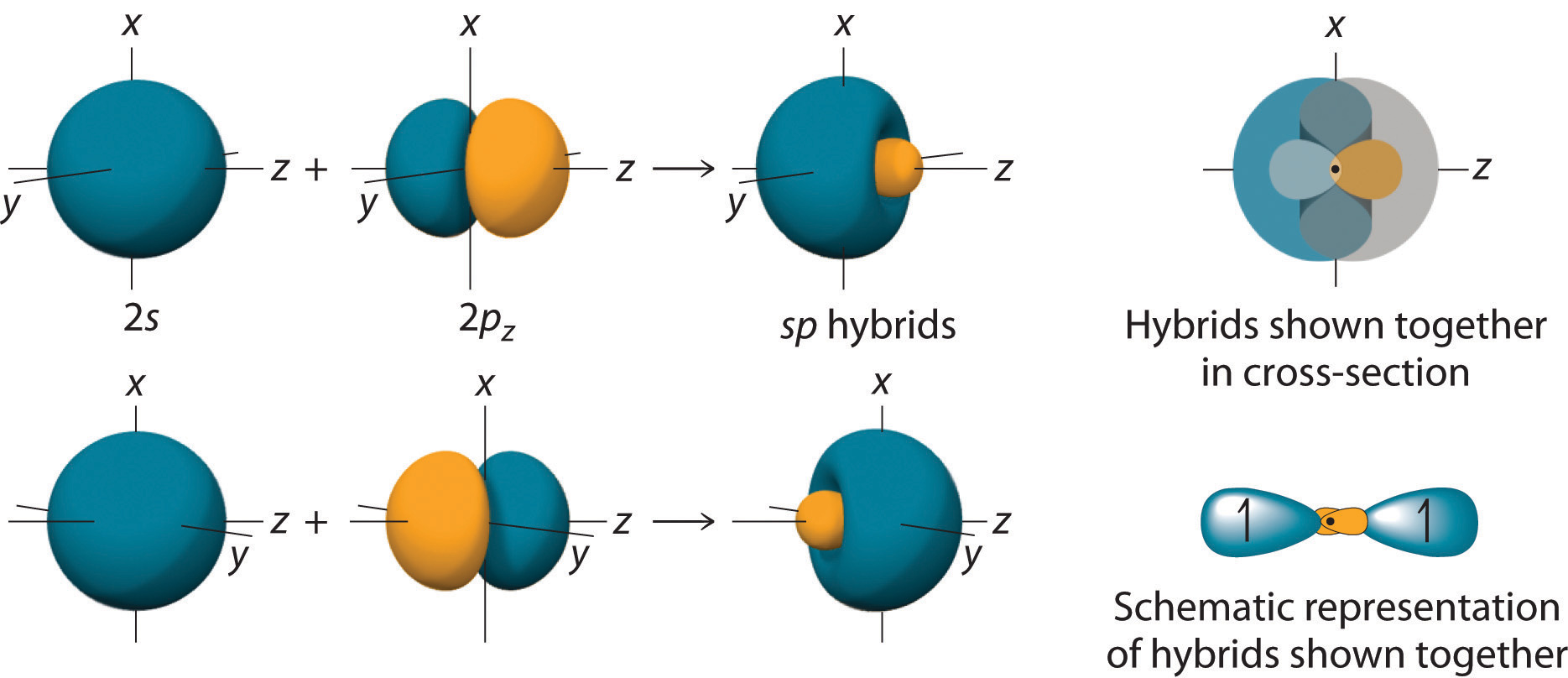
According to the valence bond theory the hybridization of central metal atom is dsp^2 for - Sarthaks eConnect | Largest Online Education Community

Valence bond theory of Coordination Compounds- Features, Hybridisation, Geometry, Examples, Limitation and FAQs of Valence bond theory.

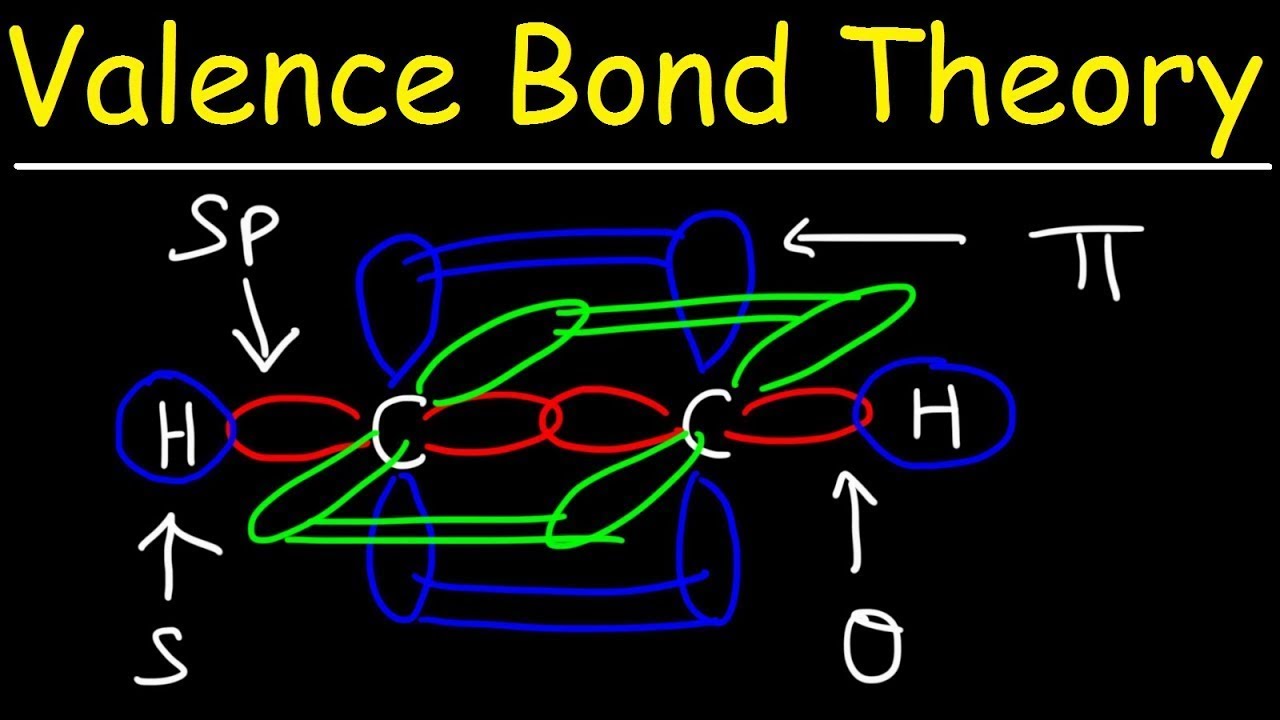



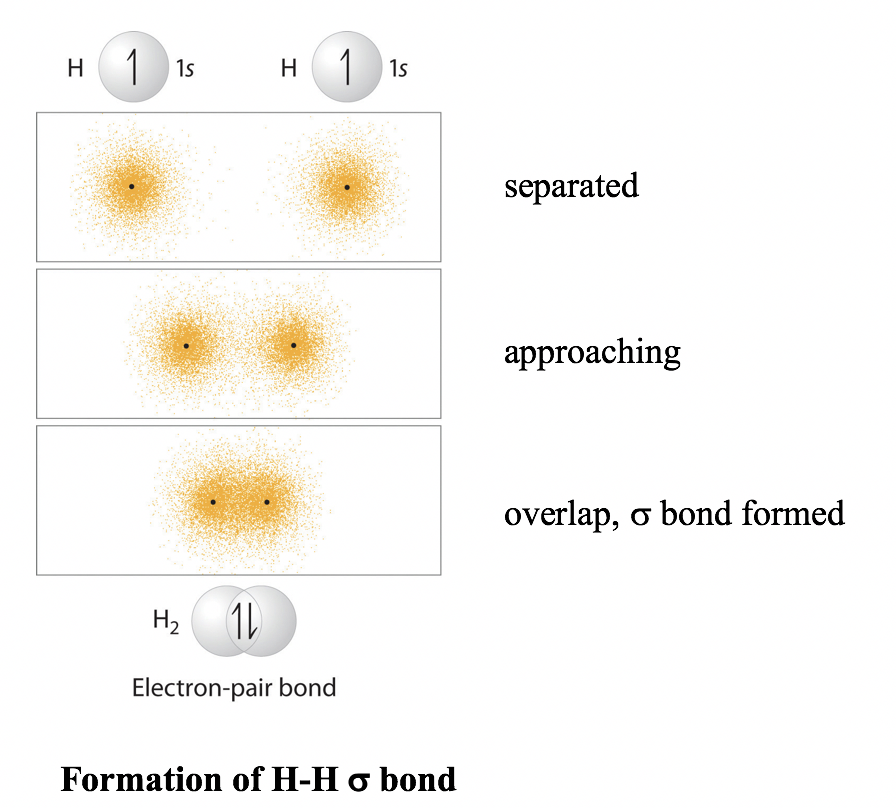


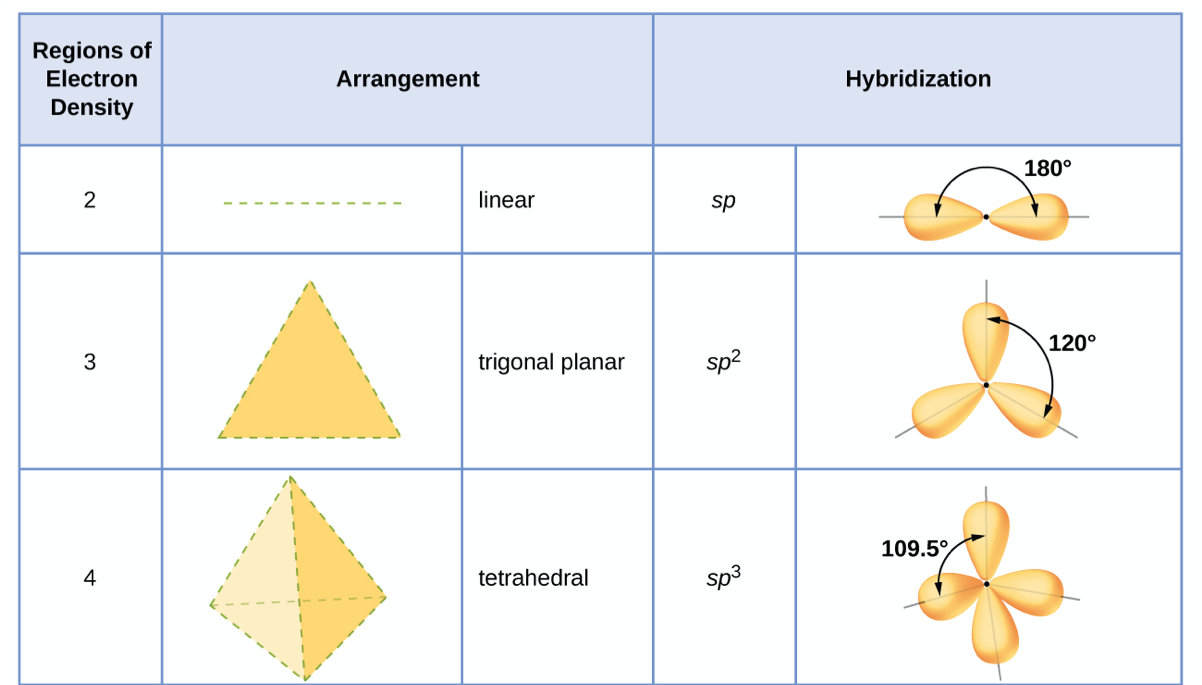

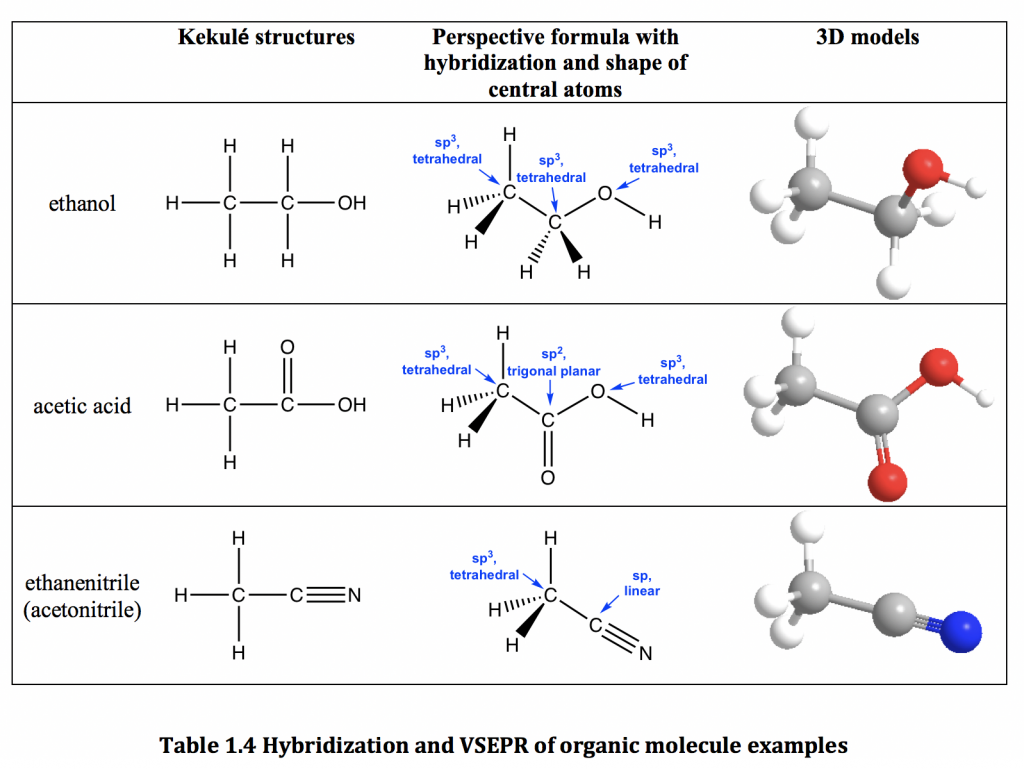
![SQP] Using Valence bond theory, explain the following in relation to SQP] Using Valence bond theory, explain the following in relation to](https://d1avenlh0i1xmr.cloudfront.net/6c2d1cd5-056b-42bc-8a63-e60054c8cb37/question-27---using-valence-bond-theory---teachoo.jpg)